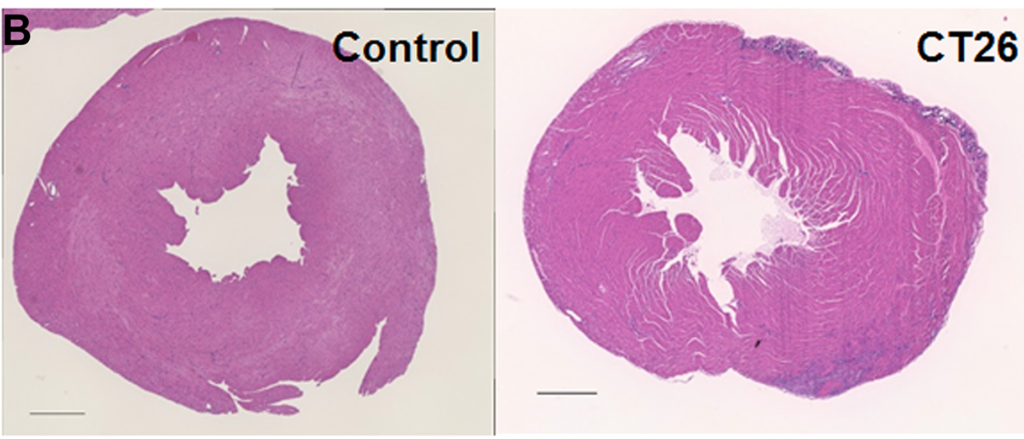
In a new research perspective, researchers discuss melatonin’s effects on cancer and the key importance of the timing of administration.

—
In the realm of cancer research, the potential of melatonin as an anti-cancer agent has garnered significant attention. Over the past 50 years, numerous studies have been conducted to investigate the effects of melatonin on tumor growth and development in mice. These studies have provided valuable insights into the complex relationship between melatonin and carcinogenesis.
In a new research perspective, researchers Vladimir N. Anisimov and Alexey G. Golubev from N.N. Petrov National Medical Research Center of Oncology wrote about the history of studies of melatonin effects on cancer in mice. Their paper was published in Oncotarget on December 12, 2023, entitled, “Melatonin and carcinogenesis in mice: the 50th anniversary of relationships.”
Early Discoveries and Controversies
In 1973, Vladimir N. Anisimov and his coauthors made a groundbreaking discovery by demonstrating the inhibitory effect of melatonin on transplantable mammary tumors in mice. This pivotal study laid the foundation for subsequent investigations into the potential anti-cancer properties of melatonin. However, early studies encountered controversies regarding the consistency of melatonin’s effects on in vivo cancer models. The lack of consistency in these studies prompted further exploration of the factors influencing melatonin’s efficacy.
Importance of Timing in Melatonin Administration
One of the crucial findings in melatonin research is the significant impact of timing in melatonin administration. Bartsch and Bartsch demonstrated that the effects of melatonin on cancer in mice depend on the time of treatment. The administration of melatonin in the morning stimulated tumor growth, while late afternoon administration inhibited it. This observation highlighted the importance of considering animal conditions and the systemic effects of melatonin when evaluating its anti-cancer properties. These systemic effects may not be evident in cell cultures or ex vivo explants.
Murine Models for Melatonin and Cancer Studies
Murine models have played a pivotal role in elucidating the effects of melatonin on various types of cancer. These models have provided valuable insights into the potential utility of melatonin in oncology. Some of the notable murine models include mice grafted with murine tumors, chemically induced tumors, spontaneous carcinogenesis in mice, transgenic HER2/neu oncogene-bearing mice, and nude mice grafted with human prostate tumors. These models have allowed researchers to evaluate not only the effects of melatonin on cancer development but also its impact on the efficacy and side effects of anticancer therapies.
Melatonin’s Effects on Spontaneous Tumor Incidence
One intriguing finding in murine studies is the effect of melatonin on spontaneous tumor incidence. Anisimov et al. showed that lifelong treatment of mice with melatonin decreased the incidence of spontaneous tumors, particularly mammary carcinomas, but only at a low concentration of melatonin in drinking water. Interestingly, this effect was not observed at a high melatonin concentration. These findings suggest that the dose of melatonin may play a crucial role in its anti-cancer effects.
Melatonin’s Role in Potentiating Cytotoxic Therapy
Another area of interest in melatonin research is its potential to enhance the efficacy of cytotoxic therapy against tumors. Panchenko et al. demonstrated that the timing of melatonin administration relative to cytotoxic drug administration significantly influenced its potentiating effect on cytotoxic therapy in HER2/neu transgenic mice. This finding highlights the importance of optimizing the timing of melatonin administration in combination with other cancer treatments.
Melatonin’s Protective Effects on Side Effects
Beyond its direct anti-cancer effects, melatonin has shown promise in alleviating the side effects of cytotoxic drugs and radiation therapy. Several murine models have demonstrated the ability of melatonin to mitigate the side effects associated with these treatments. For example, melatonin was shown to alleviate the depression syndrome in mice treated with the alkylating agent temozolomide used in brain cancer therapy. Additionally, melatonin has been found to protect against ovarian follicle depletion caused by cisplatin, a commonly used chemotherapy drug. These findings suggest that melatonin may have a broader role in cancer treatment by reducing the adverse effects of traditional therapies.
Melatonin’s Effects on Metastasis and Epithelial-Mesenchymal Transition
Metastasis is a significant challenge in cancer treatment, and melatonin has shown promise in inhibiting metastatic spread. In nude mice grafted with human gastric cancer, melatonin was found to suppress lung metastases development by inhibiting the epithelial-to-mesenchymal transition (EMT). The inhibition of EMT by melatonin has also been observed in other murine models, highlighting its potential as an anti-metastatic agent. Given the crucial role of EMT in primary cancer and metastasis development, these findings have significant implications for oncology research.
Melatonin and Inflammation
Chronic inflammation is increasingly recognized as a contributing factor in cancer development and progression. Melatonin has been found to modulate inflammatory processes in murine models. In a murine model of low-grade inflammation, melatonin inhibited EMT), suggesting a potential role in suppressing cancer-related inflammation. While the direct anti-inflammatory effects of melatonin require further investigation, these findings shed light on the multifaceted mechanisms through which melatonin may exert its anti-cancer effects.
Clinical Applications and Promising Results
The employment of melatonin in clinical settings beyond its established fields does not require licensing, making it more readily accessible for testing novel applications in cancer treatment. Promising clinical results have already been reported, such as increased overall survival in prostate cancer patients with poor prognosis after combined hormone radiation treatment. These findings highlight the potential translational impact of murine studies and underscore the importance of continued research to fully understand the clinical implications of melatonin in cancer therapy.
Conclusion
Over the past 50 years, murine models have provided valuable insights into the relationship between melatonin and carcinogenesis. These studies have shed light on the importance of timing in melatonin administration, its effects on tumor incidence and metastasis, as well as its role in potentiating cytotoxic therapy and mitigating side effects. While the precise mechanisms underlying melatonin’s anti-cancer effects require further exploration, the promising results observed in both preclinical and clinical studies warrant continued investigation. As researchers continue to unravel the complexities of melatonin’s interactions with cancer, new opportunities for therapeutic interventions may emerge, offering hope for improved cancer treatment outcomes.
“The […] main lesson being that the systemic in vivo effects of melatonin on animals may overwhelm the in vitro effects found using tissue explants or cell cultures. In particular, the timing of melatonin administration is of crucial importance for using the drug, which is freely available over [the] counter and thus needs no licensing for its applications in oncology.”
Click here to read the full research perspective in Oncotarget.
—
Oncotarget is an open-access, peer-reviewed journal that has published primarily oncology-focused research papers since 2010. These papers are available to readers (at no cost and free of subscription barriers) in a continuous publishing format at Oncotarget.com. Oncotarget is indexed/archived on MEDLINE / PMC / PubMed.
Click here to subscribe to Oncotarget publication updates.
For media inquiries, please contact media@impactjournals.com.